Bladder dysfunction can cause significant medical issues, including urinary incontinence and renal failure, yet the ability to accurately model and treat such problems has long been limited by the lack of realistic bladder models. Traditional in vitro (lab-based) models often fail to replicate the complex functions and structures of the human bladder. This gap in bladder research prompted a new development: a 3D bioprinted, biomimetic urinary bladder model that can mimic both the structure and the dynamic functions of a real human bladder.
The Need for a Better Bladder Model
The human bladder’s primary role is to store and expel urine through coordinated contractions and relaxations of the smooth muscle lining the bladder wall. Dysfunction in these processes can lead to serious medical problems, but studying bladder diseases in the lab has been challenging because current models fail to accurately replicate the human bladder’s complexity.
Conventional models often rely on either animal testing or simplistic cell cultures, neither of which capture the mechanical or structural intricacies of the bladder. To develop better treatments for bladder dysfunction, researchers need models that accurately mimic the bladder’s smooth muscle behavior, tissue environment, and biomechanical properties.
The 3D Bioprinting Approach
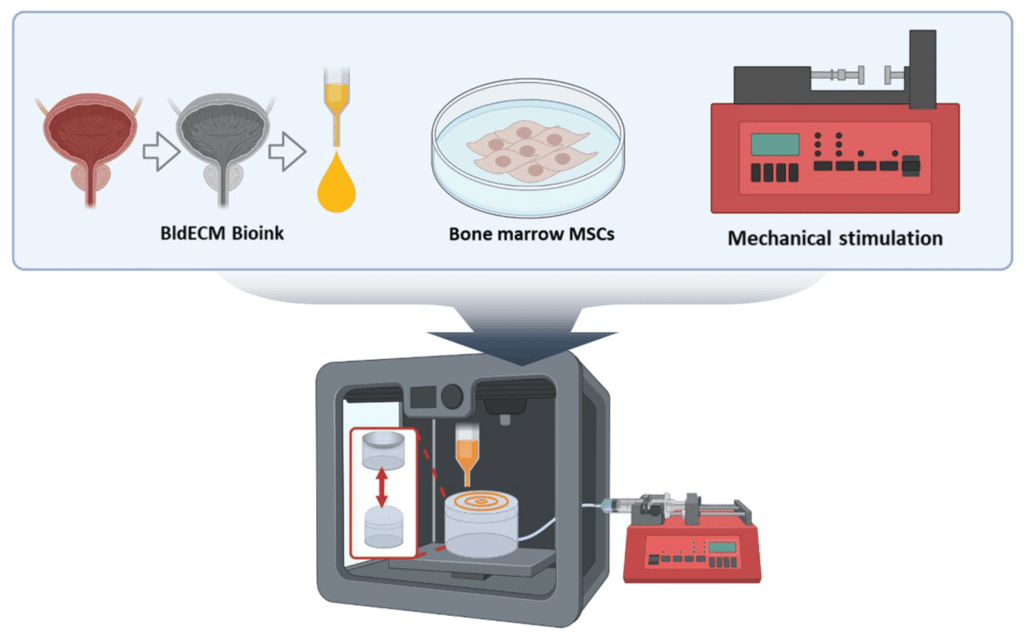
Fig. A schematic of the development of the in vitro bladder model system using 3D bioprinting and tissue-specific bioink in conjunction with the physiological stimulation system.
The study introduces a new 3D bioprinting method to create a model of the human bladder. This approach uses decellularized extracellular matrix (dECM) derived from actual bladder tissue. The dECM provides a scaffold rich in proteins, growth factors, and structural molecules that mimic the natural bladder environment. By using this dECM as “bioink,” the researchers could bioprint bladder tissue that closely mirrors real bladder smooth muscle.
The major advantage of 3D bioprinting in this case is its ability to precisely position cells and biological materials, allowing researchers to recreate the bladder’s intricate microarchitecture. The use of bladder-specific dECM improves the tissue’s biofunctionality, promoting better cell behavior like adhesion, proliferation, and myogenic (muscle) differentiation.
Contract-Release System (CRS): Mimicking Bladder Dynamics
A key innovation of this research is the introduction of a contract-release system (CRS) to simulate the bladder’s natural movements. The bladder expands and contracts during urination, and replicating this behavior is critical for developing a functional tissue model. The CRS system uses controlled air pressure to expand and contract the bioprinted bladder tissue, mimicking the physiological conditions of a real bladder.
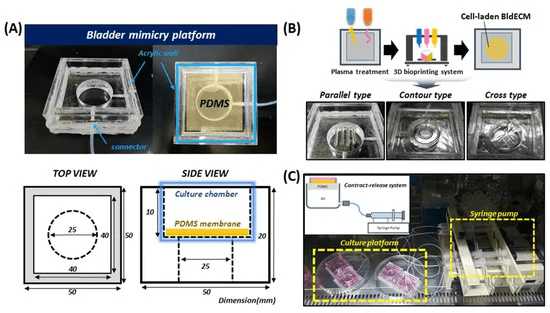
Fig. Development of a bladder mimicry platform that incorporates a contract-release system (CRS)
This mechanical stimulation not only recreates the bladder’s movement but also encourages cells to behave like bladder smooth muscle cells. Over time, the stem cells used in the bioprinted bladder tissue start to differentiate into muscle cells, thanks to the combination of the dECM and the dynamic stretching provided by the CRS.
Results and Impact on Bladder Research
The 3D bioprinted bladder tissue showed high cell viability, meaning the cells in the bioprinted model stayed healthy and continued to grow. The dynamic stretching provided by the CRS led to better differentiation of stem cells into muscle cells, making the bioprinted bladder tissue more functionally similar to a real bladder.
This biomimetic bladder model could serve as a powerful tool for researchers studying bladder diseases, testing new drugs, or even developing tissue-engineered bladder replacements. The model offers a more accurate platform for bladder research compared to traditional cell cultures or animal models. Furthermore, it opens the door for the development of personalized medicine, where treatments could be tailored based on a patient’s specific bladder condition.
Challenges and Future Directions
While this study marks a significant advancement in bladder tissue engineering, there are still challenges to overcome. The bioprinted model needs to be further developed to include more complex features, such as nerve and blood vessel integration, to fully replicate a living bladder. Additionally, scaling up the model for clinical use remains a challenge. Despite these hurdles, the development of this biomimetic bladder model represents a step forward in bladder research.
Research Work: Chae S, Kim J, Yi HG, Cho DW. 3D bioprinting of an in vitro model of a biomimetic urinary bladder with a contract-release system. Micromachines (Basel). 2022;13(2):277. doi:10.3390/mi13020277




Leave a comment