Miromatrix, a company owned by United Therapeutics, has secured Regenerative Medicine Advanced Therapy, RMAT, designation from the FDA for its miroliverELAP program. The therapy is a biofabricated external liver assist device built on decellularized porcine scaffolds seeded with human cells. It marks an important regulatory step forward, and one worth paying close attention to for anyone tracking the commercialization of engineered tissues.
What Is miroliverELAP?
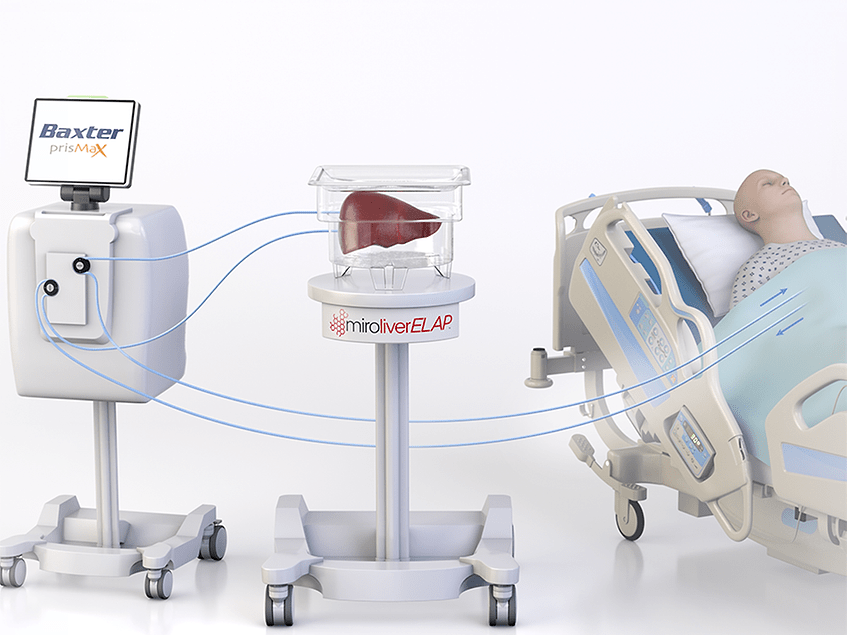
miroliverELAP, miroliver Extracorporeal Liver Assist Program, is an extracorporeal device designed to support patients in acute liver failure, bridging them to transplant or recovery. The system uses a decellularized porcine liver scaffold that is repopulated with human endothelial and hepatic cells, creating a bioengineered organ substitute capable of performing key liver functions outside the body.
Phase 1 clinical data released in early 2026 showed encouraging results. Full study outcomes are expected in the second half of 2026, which will be a critical inflection point for both the program and the broader field.
Why RMAT Matters
RMAT is the FDA’s most accelerated pathway for regenerative medicine products. It is granted to therapies that demonstrate early clinical evidence in serious or life-threatening conditions. The designation enables more frequent FDA interaction, rolling data submissions, and the potential for priority review or accelerated approval, reducing time to market.
For biofabrication, this is meaningful. RMAT signals that the FDA is willing to engage with complex engineered tissue constructs as therapeutic products, not just experimental systems. It reinforces that a decellularized xenogeneic scaffold, seeded with human cells and used in an extracorporeal circuit, can be treated as a viable clinical candidate.
The Bigger Picture at United Therapeutics
Under United Therapeutics, Miromatrix fits into a broader strategy focused on solving organ shortage at scale. Alongside lung-focused efforts and xenotransplantation programs, the addition of liver support technologies reflects a systematic, organ-by-organ approach.
The underlying thesis is straightforward: if external organ support devices can be manufactured using standardized scaffolds and human cells, they can become scalable products that do not depend on donor supply. That shifts the paradigm from scarcity-driven transplantation to engineered, repeatable therapeutics.
What to Watch
The second half of 2026 will be defining. Early results are promising, but several key questions remain:
- Durability of hepatic function over treatment duration
- Immunological response between human cells and porcine scaffolds
- Scalability and reproducibility of manufacturing
For the biofabrication industry, miroliverELAP represents a clear signal. Decellularization-based scaffold approaches are not just viable in research settings, they are reaching clinical stages with regulatory alignment.




Leave a comment