The future of endocrine cell therapy may not begin with diabetes. It may begin with a much smaller, cleaner, and more biologically focused disease: chronic hypoparathyroidism.
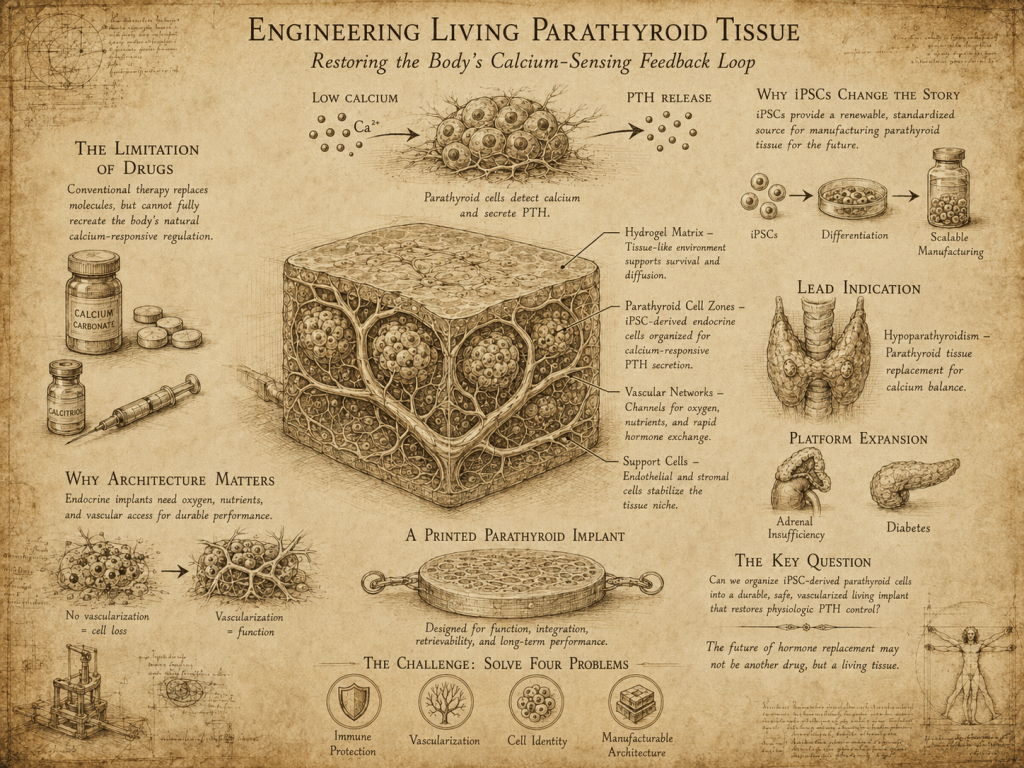
Hypoparathyroidism is a rare endocrine disorder in which the body does not produce enough parathyroid hormone, PTH. PTH is essential for regulating calcium and phosphate levels in the body. When PTH is absent or insufficient, patients can experience low blood calcium, high phosphate, neuromuscular symptoms, fatigue, brain fog, kidney complications, ectopic calcification, and long-term quality-of-life impairment. Conventional treatment usually relies on oral calcium and active vitamin D, but this approach does not restore the body’s natural calcium-sensing feedback loop. Newer PTH replacement therapies, including FDA-approved Yorvipath, have improved the field, but patients still require chronic dosing, monitoring, and lifelong disease management.
That is why hypoparathyroidism represents a compelling entry point for biofabricated hormonal tissue replacement.
The central idea is simple: instead of repeatedly dosing a hormone, build a small living tissue that can sense calcium and secrete PTH in response.
The Core Problem: Drugs Replace Hormones, Tissue Restores Feedback
Most endocrine therapies are molecular approximations of living tissue. They deliver hormones into the body, but they do not fully recreate the dynamic regulation that healthy endocrine organs provide.
This distinction matters deeply in hypoparathyroidism.
A healthy parathyroid gland is not just a source of PTH. It is a calcium-responsive sensor. When blood calcium falls, parathyroid cells increase PTH secretion. When calcium rises, PTH secretion decreases. That feedback loop helps maintain mineral balance across bone, kidney, and intestine.
Current therapies can improve calcium levels, but they do not perfectly recreate that real-time control. Calcium and vitamin D supplementation can overshoot or undershoot. Too little calcium can cause symptoms of hypocalcemia. Too much calcium or phosphate imbalance can contribute to renal complications over time. PTH analogues improve the treatment paradigm, but they still operate as administered drugs rather than self-regulating tissue.
A biofabricated parathyroid-like implant would be different.
It would aim to restore the missing endocrine function itself.
Why Hypoparathyroidism Is a Better First Target Than Diabetes
Type 1 diabetes is the largest and most validated endocrine cell therapy market, but it is also crowded, expensive, and technically demanding. Companies like Vertex, Sana, Sernova/Evotec, Aspect/Novo Nordisk, and Lilly/Sigilon have already moved aggressively into stem-cell-derived islets, immune evasion, encapsulation, and device-enabled beta-cell replacement. These programs validate the tissue-therapeutics category, but they also make diabetes a difficult first market for a new biofabrication platform.
Hypoparathyroidism is different.
It is smaller, but cleaner.
The therapeutic tissue mass is likely much smaller than what is needed for diabetes. The biology is more focused. The main hormone output is clear: PTH. The feedback signal is clear: extracellular calcium. The clinical biomarkers are direct: serum calcium, phosphate, urinary calcium, PTH, supplement burden, renal outcomes, and patient symptoms.
That makes hypoparathyroidism a strong first indication for proving a broader thesis:
Biofabricated endocrine tissue can restore physiologic hormone regulation better than chronic drug replacement.
This is a much cleaner story than trying to solve beta-cell differentiation, autoimmunity, massive cell dose, glucose responsiveness, immune protection, and high-volume manufacturing all at once.
Why iPSCs Change the Hypoparathyroidism Story
The iPSC angle is especially interesting in hypoparathyroidism because patient-specific or donor-derived parathyroid tissue is not a scalable therapeutic platform.
The body only has small parathyroid glands, and primary parathyroid tissue is not available at meaningful commercial scale. Surgical autotransplantation of parathyroid tissue can be useful in certain thyroid or parathyroid surgery settings, but that is not a broad product strategy for chronic hypoparathyroidism.
iPSCs could change that.
A renewable iPSC source could allow the generation of parathyroid-like cells at scale. These cells could be differentiated, characterized, tested for calcium-responsive PTH secretion, and incorporated into a reproducible implantable tissue format. Over time, this could support either an allogeneic off-the-shelf model or, in select cases, an autologous personalized approach.
For a first commercial product, the most realistic route is probably allogeneic iPSC-derived parathyroid tissue, not fully autologous manufacturing. Autologous iPSCs are scientifically elegant, but patient-specific manufacturing is slow, expensive, and operationally complex. In a rare disease like hypoparathyroidism, autologous manufacturing could be justified for certain high-risk or immune-complex patients, but the scalable product thesis is stronger with standardized iPSC banks.
The better framing is:
iPSCs are not just the cell source. They are the manufacturing substrate that makes parathyroid tissue replacement industrially possible.
Why Cells Alone Are Not Enough
The winning product will not simply be “PTH-producing cells.”
It will need to be a functional parathyroid microtissue.
That means the cells must survive, remain differentiated, sense calcium, secrete PTH appropriately, avoid uncontrolled growth, and function for years. They must also be placed in a body site where nutrients, oxygen, and hormone exchange are sufficient.
This is where biofabrication becomes strategically important.
Parathyroid tissue does not require the same large cell mass as pancreatic islet replacement, but it still needs the right microenvironment. The implant must support cell viability, calcium sensing, hormone release, and host integration. A poorly designed implant could contain the right cells but fail because the cells are isolated, fibrotic, oxygen-starved, or unable to respond dynamically to the bloodstream.
The product is not the cell.
The product is the cell plus architecture.
Best Architecture: A Small, Retrievable, Vascularized Parathyroid Micro-Organ
The ideal architecture for hypoparathyroidism is probably not a large organ replacement and not loose cell injection.
The best format is likely a small, retrievable, vascularized parathyroid micro-organ implant.
This could be implanted subcutaneously or intramuscularly, ideally in a location that is accessible, monitorable, and surgically retrievable. The implant would contain iPSC-derived parathyroid-like cells embedded in a hydrogel or extracellular matrix-like scaffold. It could also include endothelial cells, stromal support cells, or prevascular channels to accelerate integration with the host.
The key design features would be:
- Calcium-responsive parathyroid-like cells. The cells need to secrete PTH in response to low calcium and reduce secretion when calcium normalizes.
- Hydrogel-based tissue niche. The hydrogel should support survival, organization, diffusion, and endocrine function. It should not simply trap the cells, it should help them behave like tissue.
- Vascular support. Because PTH must enter systemic circulation and calcium must be sensed dynamically, the implant needs strong mass transfer. This may not require full perfusable vasculature at the first stage, but vascular integration should be a core design principle.
- Retrievability. This is critical. A living endocrine implant must be removable if it over-secretes, underperforms, fibroses, or raises safety concerns.
- Modular dosing. The implant should be designed as a doseable unit. Patients may need one or more microtissue units depending on body size, severity, and hormone requirement.
This is where Linton’s broader biofabrication thesis fits well. The value is not just making cells. The value is building an implant architecture that turns cells into a regulated living endocrine product.
Why Hydrogels Matter
Hydrogels are especially attractive for parathyroid tissue because they can act as a synthetic or semi-natural tissue niche.
A hydrogel can be tuned for stiffness, porosity, degradation, cell adhesion, and diffusion. It can be designed to allow calcium exchange, oxygen diffusion, and PTH release. When combined with bioprinting, the hydrogel can also organize different cell populations into defined spatial patterns.
A printed parathyroid implant could include:
- Calcium-responsive parathyroid-like endocrine zones.
- Endothelial or vascular-supporting regions.
- Stromal support cells that help stabilize the microtissue.
- Matrix cues that improve maturation and survival.
- A defined geometry that supports reproducible manufacturing and release testing.
This makes the product more sophisticated than passive encapsulation. It becomes a manufactured endocrine tissue, designed for physiological function.
Why This Could Matter Commercially
Hypoparathyroidism is rare, but the commercial logic is stronger than it first appears.
Rare endocrine diseases can support premium pricing when the disease burden is high, biomarkers are clear, and the therapy offers durable improvement. Yorvipath’s approval shows that the market recognizes the value of true PTH replacement. Reuters reported that hypoparathyroidism affects an estimated 70,000 to 90,000 people in the United States, and that analysts have projected meaningful commercial potential for Yorvipath.
That matters for a tissue therapy company because Yorvipath validates the category but also raises the bar.
A biofabricated tissue product cannot simply claim, “we replace PTH.” A drug can already do that. The tissue product has to claim something more ambitious:
We restore durable, calcium-responsive PTH physiology through a living implant.
That is the differentiated value proposition.
The product would need to show that it can reduce or eliminate dependence on oral calcium, active vitamin D, or chronic injectable PTH therapy. It would also need to show better stability of calcium and phosphate, reduced urinary calcium burden, improved symptoms, and long-term safety.
How This Matches Large Pharma Cell Therapy Programs
Large pharma is already moving toward tissue therapeutics, but mostly through diabetes.
Vertex is developing stem-cell-derived islet replacement for type 1 diabetes. Sana is pursuing hypoimmune allogeneic cell therapy. Sernova and Evotec are working on iPSC-derived beta-cell replacement using an implantable Cell Pouch strategy. Aspect Biosystems and Novo Nordisk are advancing bioprinted tissue therapeutics for diabetes and obesity. Lilly acquired Sigilon, which had encapsulated cell therapy technology relevant to endocrine diseases.
These programs validate several major themes:
- Big pharma believes endocrine cell replacement can become a real therapeutic category.
- The field is moving beyond simple cell injection.
- Architecture, immune protection, vascularization, and manufacturability are becoming central.
- Off-the-shelf cell sources are preferred for scale.
But most of this activity is concentrated in diabetes.
That leaves an opening for a company focused on rare endocrine tissue replacement, where the first product does not need to solve the massive scale and autoimmunity problems of diabetes. Hypoparathyroidism could be a beachhead that proves the biofabricated endocrine implant thesis before expanding into adrenal, thyroid, or beta-cell applications.
The strategic positioning could be:
While large pharma is building scalable beta-cell therapies for diabetes, Linton can build the architecture layer for rare endocrine tissue replacement, starting with parathyroid micro-organs for hypoparathyroidism.
The Development Path
A practical development path would start with a very focused product concept:
- Program: iPSC-derived parathyroid microtissue implant.
- Indication: chronic hypoparathyroidism inadequately controlled with standard therapy or requiring high supplement burden.
- Core function: calcium-responsive PTH secretion.
- Implant format: small, retrievable, hydrogel-based vascularizing microtissue.
- First proof point: restoration of PTH activity and calcium control in a hypoparathyroid animal model.
- Clinical biomarkers: serum calcium, phosphate, urinary calcium, PTH, calcium/vitamin D reduction, symptoms, renal safety markers.
The first milestone would not be a perfect organ. It would be a small tissue that proves regulated PTH secretion in vivo.
The second milestone would be durability.
The third would be retrievability and safety.
The fourth would be manufacturing reproducibility.
Why This Is a Better First Tissue Therapeutics Story
Hypoparathyroidism is attractive because it gives biofabrication a chance to win on architecture without needing to compete immediately against the most crowded diabetes programs.
It has:
- A clear missing hormone.
- A small target tissue.
- A direct feedback signal.
- Measurable biomarkers.
- A rare-disease commercial model.
- A validated need for better hormone replacement.
- A strong rationale for durable tissue restoration.
That combination is rare.
In diabetes, the dream is enormous, but the field is crowded and the technical burden is high. In adrenal replacement, the unmet need is compelling, but the biology is more complex. In hypoparathyroidism, the biology is focused enough to make a first product plausible.
The Big Picture
The future of hypoparathyroidism treatment may not be more pills or better injections. It may be a living implant that restores the missing endocrine tissue itself.
That is the real promise of biofabricated hormonal tissue.
A parathyroid micro-organ would not just deliver PTH. It would sense calcium, respond dynamically, integrate with the body, and provide long-term physiological control. If successful, it could establish a new category of rare endocrine tissue therapeutics.
The broader platform could then expand from parathyroid tissue to adrenal tissue, thyroid tissue, and eventually more complex metabolic tissues.
The first step should be the cleanest one:
Build a biofabricated parathyroid micro-organ for chronic hypoparathyroidism, using iPSC-derived endocrine cells in a retrievable, vascularized hydrogel architecture designed to restore calcium-responsive PTH physiology.



Leave a comment